Mycoplasma Testing Market Share, Growth, Demand and Future Revenue 2032
Mycoplasma Testing Market Size- By Technology, By Product & Services, By Distribution Channel, By Application, By End Use- Regional Outlook, Competitive Strategies and Segment Forecast to 2032
| Published: Dec-2022 | Report ID: HLCA2230 | Pages: 1 - 233 | Formats*: |
| Category : Healthcare | |||
| Report Metric | Details |
| Market size available for years | 2019-2032 |
| Base year considered | 2021 |
| Forecast period | 2022-2032 |
| Segments covered | By Technology, By Product & Services, By Distribution Channel, By Application, By End Use |
| Regions covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Companies Covered | Agilent Technologies, American Type Culture Collection, Asahi Kasei Medical Co. Ltd., Biological Industries Israel Beit Haemek Ltd., Bionique Testing Laboratories Inc., Biotools B & M Labs, Charles River Laboratories International Inc., Clongen Laboratories Inc., Creative Bioarray, Eurofins Scientific, GenBio,GeneCopoeia Inc., InvivoGen, Lonza Group Ltd., Merck KGaA, Meridian Bioscience Inc., Minerva Biolabs GmbH, Mycoplasma Experience, Nelson Laboratories Fairfield Inc., NorgenBiotek Corp., PromoCell GmbH, Roche Diagnostics, Sartorius AG, Savyon Diagnostics, ScienCell Research Laboratories Inc.,Thermo Fisher Scientific Inc. |
Based on the Application, Global Mycoplasma Testing Market is segmented as;Cell Line Testing, End-Of-Production Cell Testing, Virus Testing, Others.
1.1. Scope of the report1.2. Market segment analysis
2.1 Research data source
2.1.1 Secondary data2.1.2 Primary data2.1.3 SPER’s internal database2.1.4 Premium insight from KOL’s
2.2 Market size estimation
2.2.1 Top-down and Bottom-up approach
2.3 Data triangulation
4.1. Driver, Restraint, Opportunity and Challenges analysis
4.1.1 Drivers4.1.2 Restraints4.1.3 Opportunities4.1.4 Challenges
4.2. COVID-19 Impacts of the Global Mycoplasma Testing Market
5.1. SWOT analysis
5.1.1 Strengths5.1.2 Weaknesses5.1.3 Opportunities5.1.4 Threats
5.2. PESTEL analysis
5.2.1 Political landscape5.2.2 Economic landscape5.2.3 Social landscape5.2.4 Technological landscape5.2.5 Environmental landscape5.2.6 Legal landscape
5.3. PORTER’S five forces analysis
5.3.1 Bargaining power of suppliers5.3.2 Bargaining power of Buyers5.3.3 Threat of Substitute5.3.4 Threat of new entrant5.3.5 Competitive rivalry
5.4. Heat map analysis
6.1. Direct Assay6.2. DNA Staining6.3. ELISA6.4. Enzymatic Methods6.5. Indirect Assay6.6. PCR6.7. Others
7.1. Assays, Kits& Reagents
7.1.1. Elimination Kits7.1.2. ELISA Assays, Kits, & Reagents7.1.3. Nucleic Acid Detection Kits7.1.4. PCR Assay7.1.5. Stains7.1.6. Standards & Controls7.1.7. Others
7.2. Instruments7.3. Services
8.1. Direct Tenders8.2. Retail
9.1. Cell Line Testing9.2. End-Of-Production Cell Testing9.3. Virus Testing9.4. Others
10.1 Academic Research Institutes10.2 Cell Banks & Laboratories10.3 Clinics10.4 Community Healthcare10.5 Contract Research Organizations10.6 Hospitals and Surgical Centers10.7 Oncology Centers10.8 Pharmaceutical & Biotechnology Companies10.9 Others
11.1 North America
11.1.1. United States11.1.2. Canada11.1.3. Mexico
11.2 Europe
11.2.1. Germany11.2.2. United Kingdom11.2.3. France11.2.4. Italy11.2.5. Spain
11.2.6. Rest of Europe
11.3 Asia-Pacific
11.3.1. China11.3.2. Japan11.3.3. India11.3.4. Australia11.3.5. South Korea11.3.6. Rest of Asia-Pacific
11.4 South America
11.4.1. Brazil11.4.2. Argentina11.4.3. Rest of South America
11.5 Middle East & Africa
11.5.1. Kingdom of Saudi Arabia11.5.2. United Arab Emirates11.5.3. Rest of Middle East & Africa
12.1.1. Company details12.1.2. Financial outlook12.1.3. Product summary12.1.4. Recent developments
12.2.1. Company details12.2.2. Financial outlook12.2.3. Product summary12.2.4. Recent developments
12.3.1. Company details12.3.2. Financial outlook12.3.3. Product summary12.3.4. Recent developments
12.4.1. Company details12.4.2. Financial outlook12.4.3. Product summary12.4.4. Recent developments
12.5.1. Company details12.5.2. Financial outlook12.5.3. Product summary12.5.4. Recent developments
12.6.1. Company details12.6.2. Financial outlook12.6.3. Product summary12.6.4. Recent developments
12.7.1. Company details12.7.2. Financial outlook12.7.3. Product summary12.7.4. Recent developments
12.8.1. Company details12.8.2. Financial outlook12.8.3. Product summary12.8.4. Recent developments
12.9.1. Company details12.9.2. Financial outlook12.9.3. Product summary12.9.4. Recent developments
12.10.1. Company details12.10.2. Financial outlook12.10.3. Product summary12.10.4. Recent developments
12.11.1. Company details12.11.2. Financial outlook12.11.3. Product summary12.11.4. Recent developments
12.12.1. Company details12.12.2. Financial outlook12.12.3. Product summary12.12.4. Recent developments
12.13.1. Company details12.13.2. Financial outlook12.13.3. Product summary12.13.4. Recent developments
12.14.1. Company details12.14.2. Financial outlook12.14.3. Product summary12.14.4. Recent developments
12.15.1. Company details12.15.2. Financial outlook12.15.3. Product summary12.15.4. Recent developments
12.16.1. Company details12.16.2. Financial outlook12.16.3. Product summary12.16.4. Recent developments
12.17.1. Company details12.17.2. Financial outlook12.17.3. Product summary12.17.4. Recent developments
12.18.1. Company details12.18.2. Financial outlook12.18.3. Product summary12.18.4. Recent developments
12.19.1. Company details12.19.2. Financial outlook12.19.3. Product summary12.19.4. Recent developments
12.20.1. Company details12.20.2. Financial outlook12.20.3. Product summary12.20.4. Recent developments
12.21.1. Company details12.21.2. Financial outlook12.21.3. Product summary12.21.4. Recent developments
12.22.1. Company details12.22.2. Financial outlook12.22.3. Product summary12.22.4. Recent developments
12.23.1. Company details12.23.2. Financial outlook12.23.3. Product summary12.23.4. Recent developments
12.24.1. Company details12.24.2. Financial outlook12.24.3. Product summary12.24.4. Recent developments
12.25.1. Company details12.25.2. Financial outlook12.25.3. Product summary12.25.4. Recent developments
12.26.1. Company details12.26.2. Financial outlook12.26.3. Product summary12.26.4. Recent developments
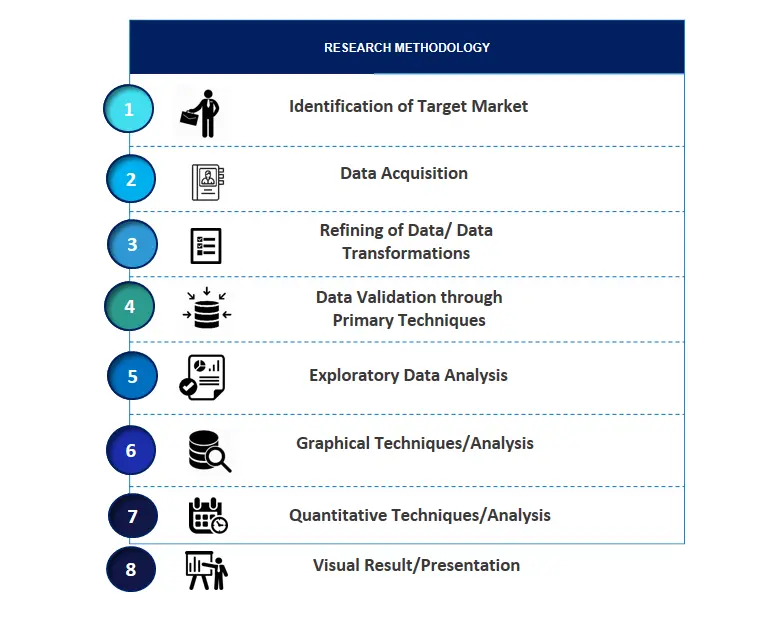
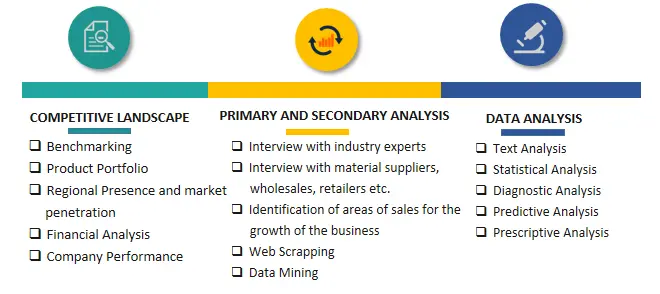
SPER Market Research’s methodology uses great emphasis on primary research to ensure that the market intelligence insights are up to date, reliable and accurate. Primary interviews are done with players involved in each phase of a supply chain to analyze the market forecasting. The secondary research method is used to help you fully understand how the future markets and the spending patterns look likes.
The report is based on in-depth qualitative and quantitative analysis of the Product Market. The quantitative analysis involves the application of various projection and sampling techniques. The qualitative analysis involves primary interviews, surveys, and vendor briefings. The data gathered as a result of these processes are validated through experts opinion. Our research methodology entails an ideal mixture of primary and secondary initiatives.
Frequently Asked Questions About This Report
PLACE AN ORDER
Year End Discount
Sample Report
Pre-Purchase Inquiry
NEED CUSTOMIZATION?
Request CustomizationCALL OR EMAIL US
100% Secure Payment






Related Reports
Our Global Clients
Our data-driven insights have influenced the strategy of 200+ reputed companies across the globe.