Viral Vector And Plasmid DNA Manufacturing Market Trends, Revenue and Future Outlook
Viral Vector and Plasmid DNA Manufacturing Market Growth, Size, Trends Analysis - By Vector Type, By Workflow, By Application, By End Use, By Disease - Regional Outlook, Competitive Strategies and Segment Forecast to 2034
| Published: Apr-2025 | Report ID: HLCA25100 | Pages: 1 - 261 | Formats*: |
| Category : Healthcare | |||

- In February 2025, Boehringer Ingelheim, in partnership with IP Group, the UK Respiratory Gene Therapy Consortium, and Oxford Biomedica, initiated the LENTICLAIRâ„¢ 1 Phase I/II trial for BI 3720931. This innovative inhaled gene therapy aims to improve outcomes for individuals with cystic fibrosis, irrespective of the specific genetic mutations causing the disease.
- In February 2025, The Parenteral Drug Association (PDA) released "Points to Consider No. 11," a comprehensive document providing guidance on the development, classification, manufacture, control, and testing of plasmids and vectors used in Advanced Therapy Medicinal Products (ATMP) production.
- In July 2024, Kaneka Eurogentec achieved a milestone by producing one kilogram of plasmid DNA from a single fermentation run, setting a new benchmark in large-scale plasmid DNA manufacturing.
- In October 2023, AGC Biologics has stated that they will expand their pDNA manufacturing facility in Germany. This is expected to help the corporation decrease the time necessary for manufacturing.
- In February 2023, BioNTech SE announced the completion of its first plasmid DNA synthesis unit in Germany. This allows the company to produce pDNA independently for clinical and commercial uses.
| Report Metric | Details |
| Market size available for years | 2021-2034 |
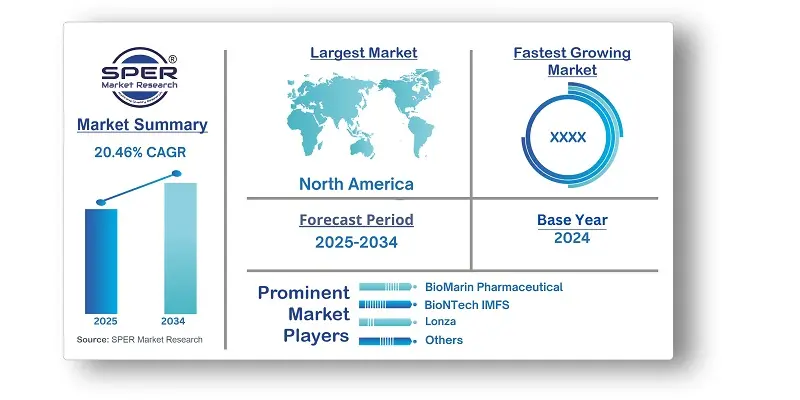
| Base year considered | 2024 |
| Forecast period | 2025-2034 |
| Segments covered | By Vector Type, By Workflow, By Application, By End Use, By Disease. |
| Regions covered | North America, Latin America, Asia-Pacific, Europe, and Middle East & Africa. |
| Companies Covered | Audentes Therapeutics, Batavia Biosciences, BioMarin Pharmaceutical, BioNTech IMFS, Catalent Inc., Cobra Biologics, Genezen laboratories, Lonza, Miltenyi Biotec, RegenxBio, Inc. |
- Global Viral Vector and Plasmid DNA Manufacturing Market Size (FY’2021-FY’2034)
- Overview of Global Viral Vector and Plasmid DNA Manufacturing Market
- Segmentation of Global Viral Vector and Plasmid DNA Manufacturing Market By Vector Type (Adenovirus, Retrovirus, Adeno-Associated Virus (AAV), Lentivirus, Plasmids and Others)
- Segmentation of Global Viral Vector and Plasmid DNA Manufacturing Market By Workflow (Upstream Manufacturing and Downstream Manufacturing)
- Segmentation of Global Viral Vector and Plasmid DNA Manufacturing Market By Application (Antisense & RNAi Therapy, Gene Therapy, Cell Therapy, Vaccinology and Research Applications)
- Segmentation of Global Viral Vector and Plasmid DNA Manufacturing Market By End Use (Pharmaceutical & Biopharmaceutical Companies and Research Institutes)
- Segmentation of Global Viral Vector and Plasmid DNA Manufacturing Market By Disease (Cancer, Genetic Disorders, Infectious Diseases and Others)
- Statistical Snap of Global Viral Vector and Plasmid DNA Manufacturing Market
- Expansion Analysis of Global Viral Vector and Plasmid DNA Manufacturing Market
- Problems and Obstacles in Global Viral Vector and Plasmid DNA Manufacturing Market
- Competitive Landscape in the Global Viral Vector and Plasmid DNA Manufacturing Market
- Details on Current Investment in Global Viral Vector and Plasmid DNA Manufacturing Market
- Competitive Analysis of Global Viral Vector and Plasmid DNA Manufacturing Market
- Prominent Players in the Global Viral Vector and Plasmid DNA Manufacturing Market
- SWOT Analysis of Global Viral Vector and Plasmid DNA Manufacturing Market
- Global Viral Vector and Plasmid DNA Manufacturing Market Future Outlook and Projections (FY’2025-FY’2034)
- Recommendations from Analyst
1.1. Scope of the report1.2. Market segment analysis
2.1. Research data source
2.1.1. Secondary Data2.1.2. Primary Data2.1.3. SPERs internal database2.1.4. Premium insight from KOLs
2.2. Market size estimation
2.2.1. Top-down and Bottom-up approach
2.3. Data triangulation
4.1. Driver, Restraint, Opportunity and Challenges analysis
4.1.1. Drivers4.1.2. Restraints4.1.3. Opportunities4.1.4. Challenges
5.1. SWOT Analysis
5.1.1. Strengths5.1.2. Weaknesses5.1.3. Opportunities5.1.4. Threats
5.2. PESTEL Analysis
5.2.1. Political Landscape5.2.2. Economic Landscape5.2.3. Social Landscape5.2.4. Technological Landscape5.2.5. Environmental Landscape5.2.6. Legal Landscape
5.3. PORTERs Five Forces
5.3.1. Bargaining power of suppliers5.3.2. Bargaining power of buyers5.3.3. Threat of Substitute5.3.4. Threat of new entrant5.3.5. Competitive rivalry
5.4. Heat Map Analysis
6.1. Global Viral Vector and Plasmid DNA Manufacturing Market Manufacturing Base Distribution, Sales Area, Interface Type6.2. Mergers & Acquisitions, Partnerships, Interface Launch, and Collaboration in Global Viral Vector and Plasmid DNA Manufacturing Market
7.1. Adenovirus7.2. Retrovirus7.3. Adeno-Associated Virus (AAV)7.4. Lentivirus7.5. Plasmids7.6. Others
8.1. Upstream Manufacturing
8.1.1. Vector Amplification & Expansion8.1.2. Vector Recovery/Harvesting
8.2. Downstream Manufacturing
8.2.1. Purification8.2.2. Fill Finish
9.1. Antisense & RNAi Therapy9.2. Gene Therapy9.3. Cell Therapy9.4. Vaccinology9.5. Research Applications
10.1. Pharmaceutical and Biopharmaceutical Companies10.2. Research Institutes
11.1. Cancer11.2. Genetic Disorders11.3. Infectious Diseases11.4. Others
12.1. Global Viral Vector and Plasmid DNA Manufacturing Market Size and Market Share
13.1. Asia-Pacific
13.1.1. Australia13.1.2. China13.1.3. India13.1.4. Japan13.1.5. South Korea13.1.6. Rest of Asia-Pacific
13.2. Europe
13.2.1. France13.2.2. Germany13.2.3. Italy13.2.4. Spain13.2.5. United Kingdom13.2.6. Rest of Europe
13.3. Middle East and Africa
13.3.1. Kingdom of Saudi Arabia13.3.2. United Arab Emirates13.3.3. Qatar13.3.4. South Africa13.3.5. Egypt13.3.6. Morocco13.3.7. Nigeria13.3.8. Rest of Middle-East and Africa
13.4. North America
13.4.1. Canada13.4.2. Mexico13.4.3. United States
13.5. Latin America
13.5.1. Argentina13.5.2. Brazil13.5.3. Rest of Latin America
14.1. Audentes Therapeutics
14.1.1. Company details14.1.2. Financial outlook14.1.3. Interface summary14.1.4. Recent developments
14.2. Batavia Biosciences
14.2.1. Company details14.2.2. Financial outlook14.2.3. Interface summary14.2.4. Recent developments
14.3. BioMarin Pharmaceutical
14.3.1. Company details14.3.2. Financial outlook14.3.3. Interface summary14.3.4. Recent developments
14.4. BioNTech IMFS
14.4.1. Company details14.4.2. Financial outlook14.4.3. Interface summary14.4.4. Recent developments
14.5. Catalent Inc.
14.5.1. Company details14.5.2. Financial outlook14.5.3. Interface summary14.5.4. Recent developments
14.6. Cobra Biologics
14.6.1. Company details14.6.2. Financial outlook14.6.3. Interface summary14.6.4. Recent developments
14.7. Genezen Laboratories
14.7.1. Company details14.7.2. Financial outlook14.7.3. Interface summary14.7.4. Recent developments
14.8. Lonza
14.8.1. Company details14.8.2. Financial outlook14.8.3. Interface summary14.8.4. Recent developments
14.9. Miltenyi Biotec
14.9.1. Company details14.9.2. Financial outlook14.9.3. Interface summary14.9.4. Recent developments
14.10. RegenxBio Inc.
14.10.1. Company details14.10.2. Financial outlook14.10.3. Interface summary14.10.4. Recent developments
14.11. Others
SPER Market Research’s methodology uses great emphasis on primary research to ensure that the market intelligence insights are up to date, reliable and accurate. Primary interviews are done with players involved in each phase of a supply chain to analyze the market forecasting. The secondary research method is used to help you fully understand how the future markets and the spending patterns look likes.
The report is based on in-depth qualitative and quantitative analysis of the Product Market. The quantitative analysis involves the application of various projection and sampling techniques. The qualitative analysis involves primary interviews, surveys, and vendor briefings. The data gathered as a result of these processes are validated through experts opinion. Our research methodology entails an ideal mixture of primary and secondary initiatives.
Frequently Asked Questions About This Report
PLACE AN ORDER
Year End Discount
Sample Report
Pre-Purchase Inquiry
NEED CUSTOMIZATION?
Request CustomizationCALL OR EMAIL US
100% Secure Payment






Related Reports
Our Global Clients
Our data-driven insights have influenced the strategy of 200+ reputed companies across the globe.